VUB study maps how brain reacts to infections
Infections can have long-term consequences
A team led by Prof Kiavash Movahedi (VUB, VIB) has mapped in detail how the immune system acts against pathogens invading the brain. The findings shed new light on host-pathogen interactions and the long-term consequences of brain infections. The results, published in the renowned journal Immunity, show how different types of immune cells are involved in the defence against brain infections. The researchers were able to show that the first line of defence is formed by brain-resident macrophages, a type of white blood cell that already lives in the healthy brain. These macrophages then recruit a large number of blood-derived macrophages that help to control the disease. These “recruited macrophages” disappear again just as quickly once the infection is resolved.
Movahedi: “The study shows the remarkable ability of the brain’s immune landscape to quickly return to its normal state. But we could also observe how certain cells remained changed for a long time after healing, having a kind of ‘memory’ of the infection. This suggests that during a lifetime, when we can suffer from numerous infections – think of the Covid-19 pandemic for example – the functionality of tissue macrophages can change. Do such earlier infections in the brain affect the likelihood of developing other neurological disorders, such as multiple sclerosis or dementia, for example? This will undoubtedly become a future hot topic in the research community.”
The brain and its defence against infections
The brain functions as a central command post that controls every process in our body. It is therefore essential that the brain can also protect itself against disease. This is partly achieved through complex barriers that restrict access to the brain. Immune cells form an additional line of defence.
Despite these various layers of defence, some pathogens still manage to penetrate, which can lead to life-threatening diseases. However, even if the infection is eventually successfully controlled and the pathogens eliminated, patients may suffer from neurological problems for years to come. It is therefore very important to better understand the dynamics of host-pathogen interaction and its long-term consequences.
The research
Prof Movahedi’s team investigated infections with Trypanosoma brucei, a dangerous microorganism that invades the brain. These single-celled parasites live in sub-Saharan Africa and can cause lethal neuropathology. Although it has long been known that these parasites infiltrate the brain, the route of infection and the nature of the host’s response remained poorly understood. The team studied the infection in mice, which, like humans, are highly susceptible to the disease.
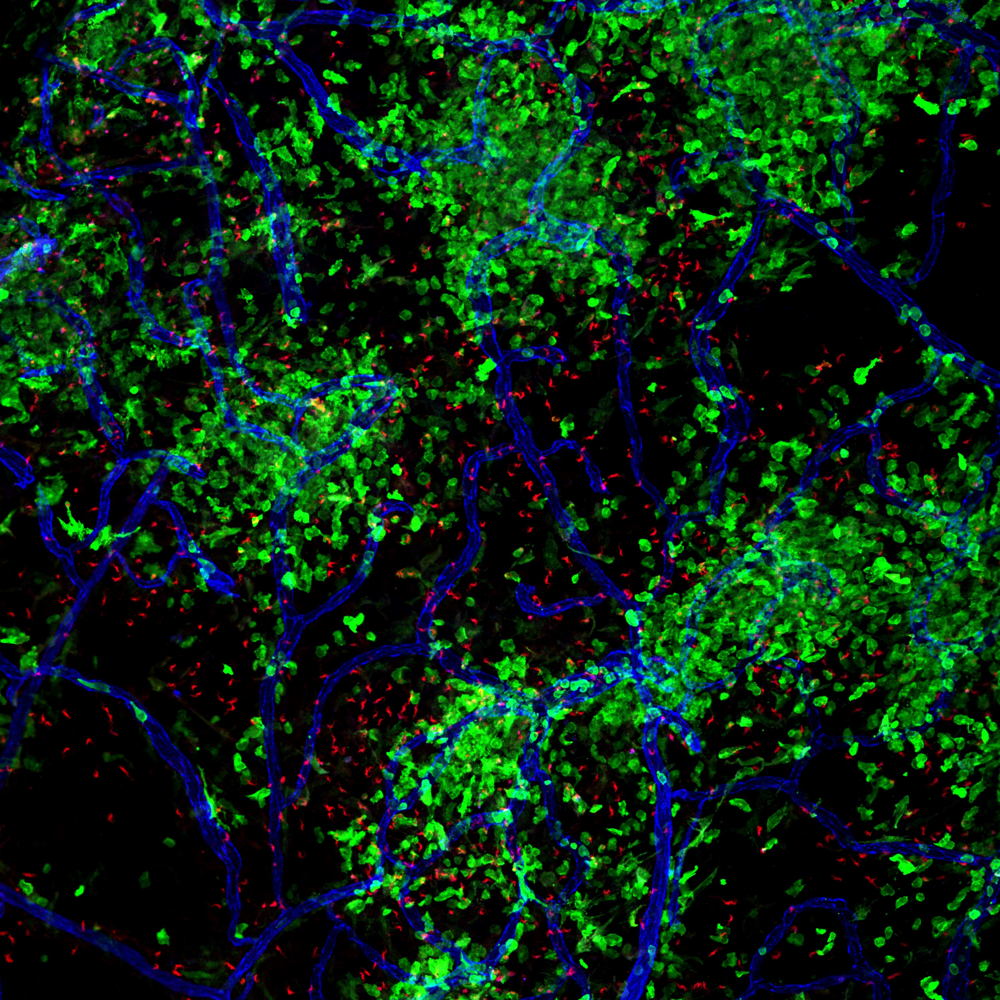
They found that the parasites first invade the brain’s border tissues, known as the meninges and the choroid plexus, and then make their way to the fluid-filled cavities and the neuronal tissue. Using several advanced technologies, the researchers also mapped the immune system’s response at great detail.
Macrophages, in particular, play a major role. They take up the first line of defence and send signals to the rest of the body to attract blood-derived immune cells to the brain. The majority of these “newly recruited” immune cells are also macrophages, but they behave differently than their resident counterparts.
“Our results place macrophages at the centre during pathogen invasion in the brain, and show how local and recruited macrophages play different roles during infection. For example, immune cells from the blood show higher expression of antimicrobial proteins. This suggests to us that they are well able to eliminate parasites,” says Karen De Vlaminck, PhD student and first author of the study.
The team then investigated what happened to all these immune cells once the disease was successfully treated.
Prof Movahedi: “We were amazed at how fast the blood-recruited macrophages were removed from the brain as soon as the parasites were eliminated. This highlights how important it is for the brain to quickly return to its normal state once an infection has been dealt with.”
Brain memory
The team found that resident macrophages in the border regions of the brain remained in a more activated state for months after the parasites were removed, suggesting that they retained some kind of “memory” of the infection. Studies in other tissues has already showed that such infection-induced memory can alter the ability of macrophages to respond to future insults.
“However, this need not be as scary as it sounds,” says De Vlaminck. “As we begin to understand the long-term impact of infections on the brain, research can start to focus on finding new therapies to address and reverse potential infection-induced dysfunctions.”
Publication in Immunity: "Differential plasticity and fate of brain-resident and recruited macrophages during the onset and resolution of neuroinflammation"
DOI10.1016/j.immuni.2022.09.005
Contact:
Kiavash Movahedi, Laboratory of Molecular and Cellular Therapy
kiavash.movahedi@vub.be
+32 473 188 213

.jpg)